Bayer Recalls 6 mL Size Afrin Original Nasal Spray Bottles Due to Risk of Serious Injury or Illness from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging
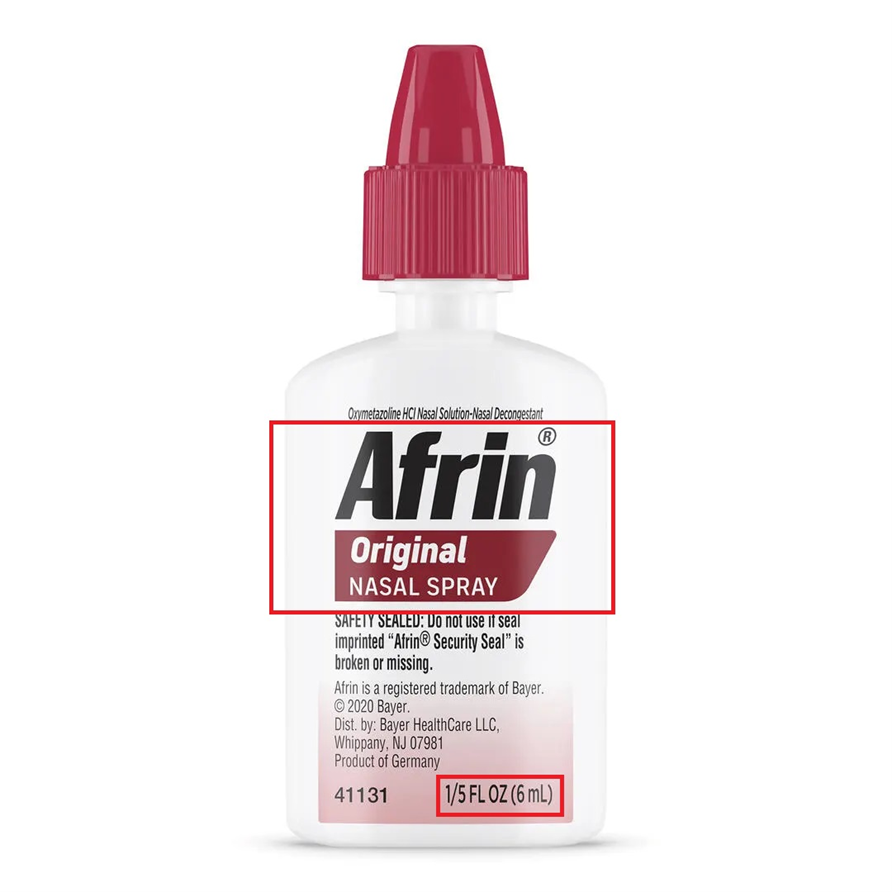
This recall involves unexpired Travel Size Afrin® Original Nasal Spray 6 mL bottles, with Lot numbers 230361, 240822, 241198, 250066, 250152, 250646, and 250831. These travel size bottles have "Afrin® Original Nasal Spray" and "1/5 FL OZ (6 mL)" printed on a label located on the front of the bottle. It is a six-digit number, followed by the expiration date in following format: "YYYYMMM". No other sizes of Afrin are...
Official details
- Category
- Consumer product recall
- Company or vendor
- Bayer HealthCare LLC, of Whippany, New Jersey; Retailers such as convenience stores and travel hubs including airports from September 2024 to April 2026 for about $7 to $9.
- Affected product
- Travel Size Afrin® Original Nasal Spray 6 mL Bottles
- Risk or reason
- The 6 mL nasal sprays contain an imidazoline, which must be in child-resistant packaging or meet the labeling requirements for non-complying packaging, as required by the Poison Prevention Packaging Act. The 6 mL nasal spray's packaging is not child-resistant nor bears the required labeling statement, posing a risk of serious injury or illness from poisoning, if the contents are swallowed by young children.
- Remedy or action
- Consumers should immediately secure the recalled bottles out of sight and reach of children and go to the webform posted on www.livewell.bayer.com/afrin-original-spray-recall to begin a request for a refund. Consumers will be asked to take and submit a photo of the product before disposing of it in order to receive a refund.
- Official ID
- 26455